|
Although it has been the subject of much scrutiny, the washing procedure
which has been developed through the years is actually quite good. There
are many techniques used in washing a hair sample prior to testing for
the presence of cocaine. These can include washing with shampoos, aqueous
solutions like buffers or water, and organic solvents like methylene
chloride, surfacants, and acetone. Care must be taken when washing a
hair sample to ensure that ingested (or internal) cocaine is not washed
out, but that external contamination is removed.[5,13] Aqueous buffers
and organic solvents can solubilize drugs from hair and make concentrations
in hair appear lower than they actually are, and therefore, are not
recommended.
There
are three domains in hair called the surface domain, the accessible
domain, and the inaccessible domain. The surface domain is the surface
of the hair and is where most external contamination occurs. The surface
domain can be washed with non-swelling agents like ethanol for sufficient
removal of external contamination. The accessible domain is the middle
portion of the hair shaft and it is slightly vulnerable to external
contamination. The accessible domain may be freed of external contamination
using swelling solvents which open up the outside matrix of the hair
enough to get some of the external contamination in the accessible domain
out. Water is a swelling solvent which is often used for this purpose.
The inaccessible domain is by far the largest domain and is the portion
of hair in the middle of the hair shaft. It is nearly impossible for
drugs to contaminate this part of the hair externally, although soaking
hair in a drug solution has been shown to cause minimal contamination
of the inaccessible domain. Neither ethanol nor water disturbs the inaccessible
domain of the hair, so ingested cocaine remains in the hair while external
contamination is removed.[13]
The delicate
balance between removing all of the external contamination and not removing
ingested cocaine is reached through two procedures. The procedures are
called truncated washing and extended washing. The truncated washing
procedure is used in screening and in work place testing because it
is much less time consuming and more practical. The number and duration
of the washes is fixed for all hair samples at a value which cleans
the average sample sufficiently. The procedure includes one fifteen
minute wash in ethanol followed by three thirty minute washes with phosphate
buffer. The extended wash is used in criminal cases and other cases
where accurate results carry much importance. The extended procedure
involves taking samples of each washing and testing them for drug levels
using radioimmuno assay until the washing level reaches a plateau when
the hair is assumed to be washed completely. The plateau is determined
by the ratio of the total amount of drug in the hair wash to the total
amount removed by washings. If the ratio is greater than 0.25 the washing
is complete. The results of the extended wash procedure are best seen
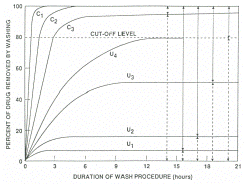
by graphing the concentrations of cocaine after each wash. In Figure
1,
(Click on the image to see an enlarged Figure in a separate browser
window.)
curve
C1 refers to a sample which was contaminated only on the surface domain.
C2 shows more severe external contamination in which the accessible
interior domain was affected by the contamination. C3 is a sample which
was so severely contaminated externally that the inaccessible interior
domain was moderately penetrated. It is believed that exposure as severe
as that in C3 would result in a positive urine test as well. The curves
labeled U in the figure represent positive samples, sample with a ratio
lower than the cut-off level. The differences in the curves results
from differences in porosity and thickness of the hair used in the testing.
The curve U1 is of a very thick and non-porous sample where U3 is a
very porous sample due to chemical cosmetic treatment. Curve U4 is a
very rare case of extremely damaged hair which may skew results because
internal contamination is removed during the washes. This can be remedied
by using less harsh solvents during the washing.[13]
Different
solvents are used in these procedures depending upon the type of hair
in the sample. Porous hair is treated with ethanol and water while nonporous
hair is treated with phosphate buffer. The porosity of the sample can
be determined through staining with methylene blue. This staining involves
an extra rinsing step and examination with a microscope. Porosity can
be caused by cosmetic treatment, race differences, or bad preservation
of hair. The conclusions reached from many studies on the washing procedure
have determined that washing is necessary and have suggested examples
for standardization.[13]
After
the hair sample is washed, it is necessary to extract the drugs and
their metabolites from the hair sample. The sample must be put through
these procedures without being degraded, hydrolyzed, or otherwise altered.
Suggested methods of treatment for extracting hair are: treatment with
alkali, acids, enzymes, or other organic solvents. Some specific substances
used in these extractions are tris buffer, dithiothreitol, sodium dodecyl
sulfate, proteinase K, and phosphate buffers. If the extraction occurs
near a pH value of 6.2, the cocaine is safe from conversions into hydrolysis
products.[13] The treatment with enzymes seems to be the safest way
to extract hair using wet chemistry because it does not compromise the
sample in any way.[14] The acid and base extractions are known to hydrolyze
cocaine. Extraction also removes melanin which is a major culprit of
race biases in hair testing.
The
extraction procedure is the step in which the drug is released from
the hair matrix, purified, and concentrated. A good extraction method
must remove all of the drug from the hair and not degrade the sample.[18]
Methanol extraction, acid/base extraction, and enzymatic extraction
have all been studied. Methanol extraction is surprisingly good for
many (but not all) drugs, while acids and bases may cause hydrolysis,
the products of which may be identical to the metabolites. Enzymatic
extraction works well with good recoveries, but involves the use of
unpleasant thiols.[14] Extraction by the liquid methods is labor intensive,
time-consuming, and is therefore expensive. An extraction technique
which is popular and seems a likely candidate for standardization is
supercritical fluid extraction using CO2. Supercritical fluid extraction
is easy, fast, mild, and does not degrade the hair sample. The extraction
conditions using supercitical CO2 can be changed easily by changing
the pressure, temperature, or modifier. The hair sample is first "rinsed"
with supercritical CO2 which does not remove the cocaine from the hair
matrix. A modifier solution is then added to the CO2 to allow for extraction
of the cocaine from the hair matrix. A good modifier has been found
to be CO2/MeOH/Et3N/H2O.[19] The desorption of cocaine from the matrix
is the rate limiting step in the extraction, so the extraction is very
dependent on the hair matrix. The modifier works because the triethylammonium
cation competes with cocaine for the negative binding sites in the hair.
The addition of water helps the extraction by facilitating formation
of the triethylammonium cation and by swelling the hair and making the
binding sites more accessible to the solvent. Supercritical fluid extraction
is not a good technique to recover benzoylecgonine because benzoylecgonine
has additional "binding sites" that may also be changed. Since this
compound is present in very small amounts in the first place, a good
extraction technique for benzoylecgonine is essential if the metabolite
is to be used as an internal contamination marker.
Supercritical
fluid extraction gives excellent recoveries of cocaine and its metabolites,
is simple, rapid, environmentally sound, and can be applied to many
types of drugs.[12] Supercritical fluid extraction can also be easily
coupled on line with many chromatographic techniques to avoid contamination
and to give better detection.[14] Liquid-liquid and solid phase extraction
have been investigated significantly, but have been found inferior to
supercritical fluid extraction.
A variety
of detection methods are also used in drug analysis in hair. Most of
these techniques are chromatographic, because chromatography gives the
required sensitivity and selectivity. The first technique used though
was not a chromatographic technique. It was radioimmuno assy, which
is still used in some cases for screening because it meets the needs
for sensitivity, throughput, and simplicity needed in these instances.[5]
All results from this technique must be verified through GC-MS testing
though and this technique involves using radioactive reagents. Immunoassay
can only screen for one class of drugs at a time which often makes multiple
runs necessary.[16] The specificity of the radioimmuno assay technique
is limited to classes of drugs and does not allow specific drug identification.
Quantification is not possible using this technique either.[6]
More
popular techniques include TLC, HPLC, LC, GC, and GC-MS. Thin layer
chromatography (TLC) techniques have limited mass sensitivity and are
improved by using high performance thin layer chromatography (HPTLC)
with dansyl derivation. TLC shows good mass and quantitation limits.
Flourescence detection is usually used with TLC detection of cocaine.[5]
High performance liquid chromatography (HPLC) is also useful for detection
of cocaine in hair samples. It is more suitable than other chromatographic
techniques for biological extracts. Sensitivity and selectivity can
be excellent with electrochemical and flourescence detectors. It can
handle high work loads and is easily automated. It shows good correlation
to results from radioimmunological assay testing.[5] It is rarely used
in hair analysis though because it uses too much solvent and mass sensitivity
could be improved by using other techniques.[9] Liquid chromatography
(LC) has played a very minor role in cocaine analysis and warrants only
a brief mention. The last purely chromatographic technique used is gas
chromatography (GC). GC is less useful than TLC or HPLC. Many products
are found and the spectra are hard to interpret. GC shows poor sensitivity
and poor selectivity. Unless coupled with a mass spectrometer, GC is
not a viable tool in drug detection in hair.[6,5]
Gas chromatography-mass
spectrometry (GC-MS) is considered the gold standard in hair testing.
Almost every positive result found is confirmed by GC-MS and it is usually
the only test which is accepted in the courts. GC-MS is the tool of
choice because it has good sensitivity, selectivity, specificity, a
high degree of standardization, sample throughput, and instrument ruggedness.
The GC-MS is usually run in the electron ionization-single ion monitoring
(EI-SIMs) mode, because this is a good compromise between selectivity
and sensitivity. The two ion mode can be used to looking for multiple
drugs at the same time. Chemical ionization (CI) is more sensitive than
EI and is becoming more popular, but EI is still the preferred ionization
method.[5] Advantages of GC-MS over other techniques include a minimal
amount of sample use and the ability to screen for the seven most important
drugs during one run.[16] The one criticism of GC-MS is that it requires
derivitization of the extracted compounds and therefore introduces an
additional step with the liabilities associated.[9] |